IGCSE Chemistry Notes – Arrangement of Elements
Topic 1
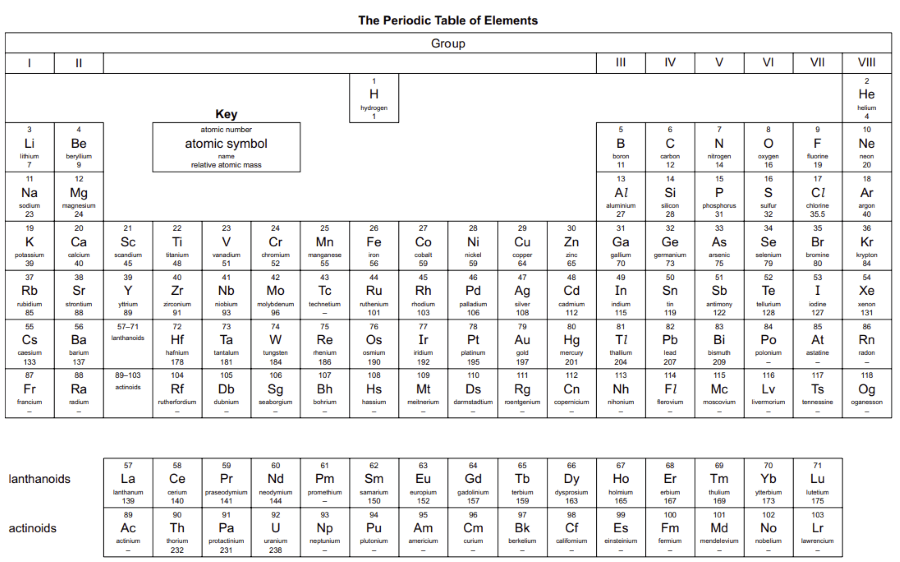
Describe the Periodic Table as an arrangement of elements in periods and groups and in order of increasing proton number / atomic number
Definition
The Periodic Table is a table where elements are arranged in order of increasing atomic number (proton number) .
Atomic number = number of protons in the nucleus of an atom.
Periods
Horizontal rows in the Periodic Table There are 7 periods
Period number = number of occupied electron shells.
Sodium (Na) / Period 3 / 3 electron shells
Groups
Vertical columns in the Periodic Table Elements in the same group have similar chemical properties
For main group elements, group number = number of electrons in the outer shell (valence electrons) .
Element
Group
Outer electrons
Lithium
1
1
Magnesium
2
2
Aluminium
3
3
Chlorine
7
7
Elements are arranged so that similar elements appear in the same column (group) . This shows repeating patterns in their chemical and physical properties .
Topic 2
Describe the change from metallic to non-metallic character across a period
Main trend
Across a period, metallic character decreases and non-metallic character increases .
Na , Mg , Al , Si , P , S , Cl , Ar
Trend explanation
Left side
Right side
Metals
Non-metals
Lose electrons easily
Gain electrons easily
Form positive ions
Form negative ions
Good conductors
Poor conductors
Nuclear charge increases Atoms hold electrons more strongly It becomes harder to lose electrons It becomes easier to gain electrons
Topic 3
Describe the relationship between group number and the charge of the ions formed from elements in that group
General rule
Main group elements form ions to obtain a full outer electron shell (noble gas configuration) .
Group
Ion formed
Charge
1
Lose 1 electron
+1
2
Lose 2 electrons
+2
3
Lose 3 electrons
+3
5
Gain 3 electrons
-3
6
Gain 2 electrons
-2
7
Gain 1 electron
-1
0
Do not form ions
stable
Sodium (Group 1)
Na -> Na+ + e-
Forms a +1 ion .
Magnesium (Group 2)
Mg -> Mg2+ + 2e-
Forms a +2 ion .
Chlorine (Group 7)
Cl + e- -> Cl-
Forms a -1 ion .
Topic 4
Explain similarities in the chemical properties of elements in the same group of the Periodic Table in terms of their electronic configuration
Elements in the same group have similar chemical properties because they have the same number of outer shell electrons .
Example: Group 1 (Alkali Metals)
Element
Electron configuration
Lithium
2,1
Sodium
2,8,1
Potassium
2,8,8,1
All have 1 outer electron All form +1 ions React vigorously with water Form alkaline solutions
Example: Group 7 (Halogens)
Element
Electron configuration
Fluorine
2,7
Chlorine
2,8,7
Bromine
2,8,18,7
All have 7 outer electrons All gain 1 electron All form -1 ions React with metals to form salts
Na + Cl ? NaCl
Topic 5
Explain how the position of an element in the Periodic Table can be used to predict its properties
From the group number
Number of outer electrons Type of ion formed Type of bonding Reactivity
From the period number
Number of electron shells Approximate atomic size
Example prediction: Group 2, Period 3
2 outer electrons Forms a 2+ ion Metal Reacts with acids Forms ionic compounds
This describes Magnesium (Mg) .
Example: Element in Group 7
Non-metal Forms -1 ions Reactive Forms salts with metals
Topic 6
Supplement – Identify trends in groups, given information about the elements
Example: Group 1 (Alkali Metals)
Property
Trend down group
Reactivity
Increases
Melting point
Decreases
Atomic size
Increases
Outer electron is further from the nucleus It becomes easier to remove
Example: Group 7 (Halogens)
Property
Trend down group
Reactivity
Decreases
Melting point
Increases
Colour
Darker
Exam Summary
Summary of key exam points
Periodic Table is arranged by increasing atomic number Periods = rows, Groups = columns Group number = number of outer electrons Across a period, metallic character decreases Group number helps predict ion charge Elements in the same group have similar chemical properties Position helps predict reactivity and bonding
Write a public review