Describe metallic bonding as the electrostatic attraction between the positive ions in a giant metallic lattice and a ‘sea’ of delocalised electrons
- Metallic bonding is a third type of giant structure.
- The metal atoms are closely packed together in a regular arrangement.
- As they are very close to each other, the valence electrons tend to drift away from the atoms.
- Thus a sea of delocalised ( mobile) electrons is formed surrounding the positive metal ions.
- The positively charged metal ions are held together by their strong attraction to the mobile electrons that keep moving between the ions.
- This is metallic bonding.
- The electrostatic attraction between the metal ions and the electrons exists in all directions.
Explain in terms of structure and bonding the properties of metals:
- (a) good electrical conductivity
- (b) malleability and ductility
- Metals have good electrical conductivity
- Reason: When a voltage is applied, the delocalised electrons move through the metal lattice towards the positive pole of the cell or power pack.
- But if the vibrations of atoms becomes faster due to high temperature , The electrons will not be able to move easily through the lattice.
- That is why the electrical conductivity of a lattice decreases with an increase in temperature.b) malleability and ductility
(b)Metals are malleable and ductile.
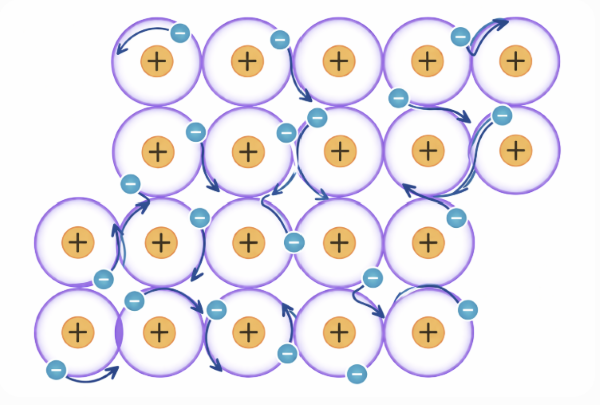
- The positive ions in a metal are arranged regularly in layers.
- When a force is applied, the layers can slide over each other.
- In a metallic bond, the attractive forces between the metallic ions and the electrons exist in all directions.
- So when the layers slide, new bonds can easily form.
- This leaves the metal with a different shape.


.png)
Write a public review