1. Reduction
Reduction is the loss of oxygen
It is also an decrease in the oxidation state
It is a gain of hydrogen
It is a gain of electrons
2. Oxidation
Oxidation is gain of oxygen.
It is also an increase in the oxidation state .
It is a loss of hydrogen
It is a loss of electrons
Examples
Reduction/Oxidation in terms of oxygen
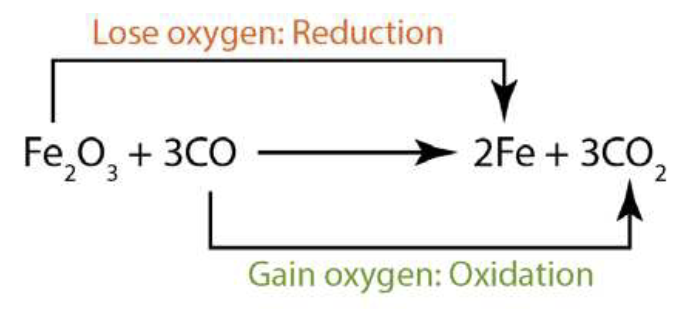
Fe2O3 loses oxygen and gets reduced to Fe.
(Reduction)
Fe2O3 + 3CO --> 2Fe + 3CO2
CO accepts oxygen and gets oxidised to CO2
(Oxidation)
Reduction/ Oxidation in terms of electrons
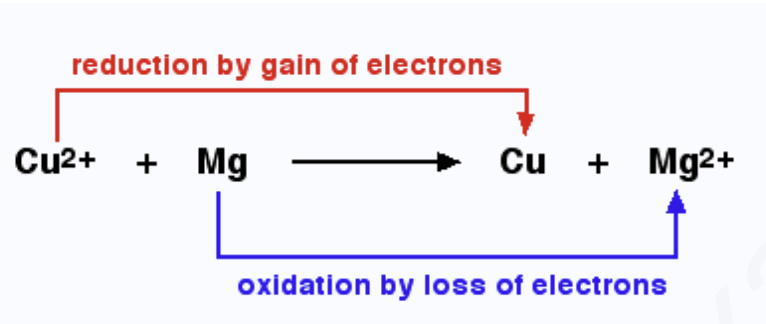
Mg loses 2 electrons to form Mg2+ ions.
(Mg gets oxidised to Mg2+ ions)
At the same time:
Cu2+ accepts these 2 electrons (2e?) and gets reduced to Cu atoms
Cu2+ + (2e-) -> Cu
(Cu2+ ions get reduced to Cu atoms)
Reduction/ Oxidation in terms of hydrogen
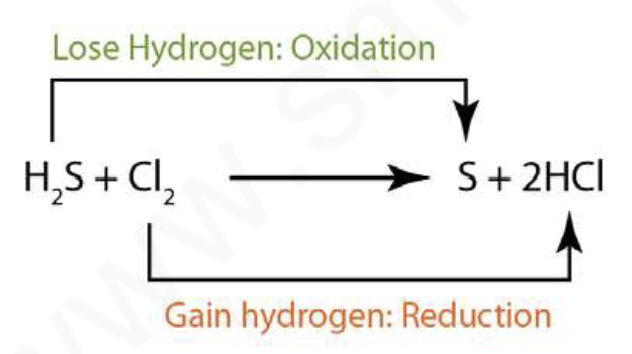
H2S loses hydrogen and gets oxidised to S.
( H2S is getting oxidised)
H2S + Cl2 -> S + 2HCl
At the same time Cl2 accepts hydrogen and forms HCl (Cl2 is getting reduced)
Reduction/ Oxidation in terms of oxidation state
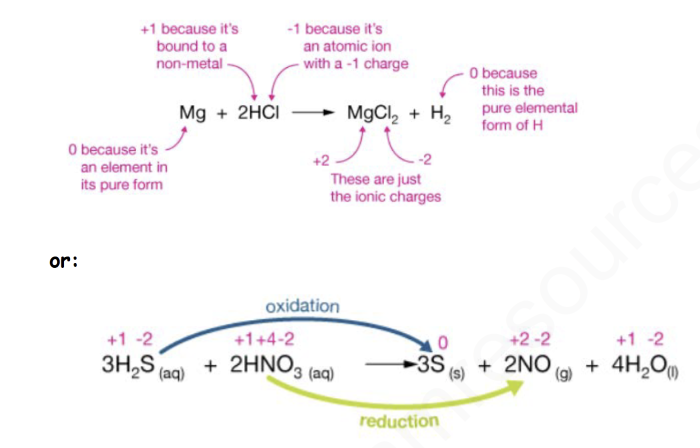
3H2S (aq) + 2HNO3 (aq) -> 3S (s) + 2NO + 4H2O
Rules for oxidation state:
- The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Thus, the atoms in O2, O3, P4, S8, and aluminum metal all have an oxidation number of 0.
- The metals in Group IA form compounds (such as Li3N and Na2S) in which the metal atom has an oxidation number of +1.
- The elements in Group IIA form compounds (such as Mg3N2 and CaCO3) in which the metal atom has a +2 oxidation number.
- Oxygen usually has an oxidation number of -2. Exceptions include molecules and polyatomic ions that contain O-O bonds, such as O2, O3, H2O2, and the O22- ion.
- The elements in Group VIIA often form compounds (such as AlF3, HCl, and ZnBr2) in which the nonmetal has a -1 oxidation number.
- The sum of the oxidation numbers in a neutral compound is zero.
H2O: 2(+1) + (-2) = 0
- The oxidation number of hydrogen is +1 when it is combined with a nonmetal as in CH4, NH3, H2O, and HCl.
- The oxidation number of hydrogen is -1 when it is combined with a metal as in. LiH, NaH, CaH2, and LiAlH4.
- The sum of the oxidation numbers in a polyatomic ion is equal to the charge on the ion. The oxidation number of the sulfur atom in the SO42- ion must be +6, for example, because the sum of the oxidation numbers of the atoms in this ion must equal -2.
SO42-: (+6) + 4(-2) = -2
- Elements toward the bottom left corner of the periodic table are more likely to have positive oxidation numbers than those toward the upper right corner of the table. Sulfur has a positive oxidation number in SO2, for example, because it is below oxygen in the periodic table.
SO2: (+4) + 2(-2) = 0
- For elements that form more than one ion, the oxidation state can be worked out from the formula of its oxide.
Reducing agent
Is a substance that reduces another substance during a redox reaction.
Potassium iodide in acidic solution is a good reducing agent. When it reduces a substance, its colour changes from colourless to brown.
Other reducing agents are:
- Carbon monoxide( CO)
- Carbon(C)
- Ammonia(NH3)
- Sulfur dioxide(SO2)
3. Oxidising agent:
Is a substance that oxidises another substance during a redox reaction.
Potassium manganate (VII) in acidic solution is a good oxidising agent. When it oxidises a substance its colour changes from purple to colourless.
Common oxidising agents:
- Halogens: Cl2,Br2, I2 etc..
- Oxygen [O2]
- Hydrogen peroxide [H2O2]
- Potassium dichromate (VII) [K2Cr2O7]
4. Redox
A reaction in which reduction and oxidation take place simultaneously is said to a redox reaction
Note:
- Oxidation state is always written in the name of compounds having more than one oxidation state.
- A compound of a transition element must include its oxidation state; Example iron oxide. If it is not mentioned then we will not come to know whether the compound is Fe(II) oxide or Fe(III) oxide as iron can have more than one oxidation state.




Write a public review