Core :
EXTENDED/SUPPLEMENT
IONS AND IONIC BONDS
Formation of sodium chloride:
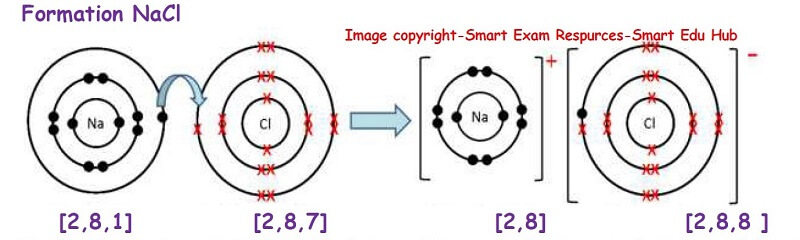
In the formation of sodium chlorine, sodium atom loses one electron and becomes a +vely charged cation. The chlorine atom accepts this electron and forms a negatively charged ion called the anion. Thus by doing so both the ions have a stable electronic structure which is the same as the noble gas. So a stable electronic structure has been formed.
Formation of calcium chloride:
Ionic and covalent-Compounds-A comparison
The above video is a preview of the actual revision notes available as a part of the paid membership. All learning resources in the form of pdfs that are available on the website are 100% printable and downloadable.
| Covalent | Ionic | |
|---|---|---|
| Formed between | Non -metals | Metals and non-metals |
| Melting points and boiling points | Low melting and boiling points because the intermolecular attractive forces are very weak. Exceptions are: SiO2 with a high melting point | High melting and boiling points because of strong electrostatic forces between the ions in the giant lattice |
| Solubility | Insoluble in water ( Exceptions: sugar and amino acids-water soluble) | Soluble in water because the water molecules are able to separate the ions from one another and keep them in the solution. |
| Electrical conductivity | Do not conduct electricity because they have no ions. Hydrogen chloride gas, a covalent compound reacts with water to form HCl acid which splits up into ions. | They conduct electricity in the molten or aqueous form due to the presence of mobile ions. |
Click the highlighted blanks to reveal the correct answer.
Unlock the complete range of solved past exam papers along with mock exams, revision notes, timed quizzes, interactive flipbooks and many more exam preparation resources.
Unlock Complete Resources
Write a public review