We use cookies to ensure that we give you the best experience on our website. We adhere to the GDPR and EU laws and we will not share your personal information with or sell it to third-party marketers. If you continue to use this site we will assume that you are happy with it. Our privacy policy
Cookie policy Download PDF
Download PDF
Syllabus Objectives:
Core:
STRUCTURE OF AN ATOM:
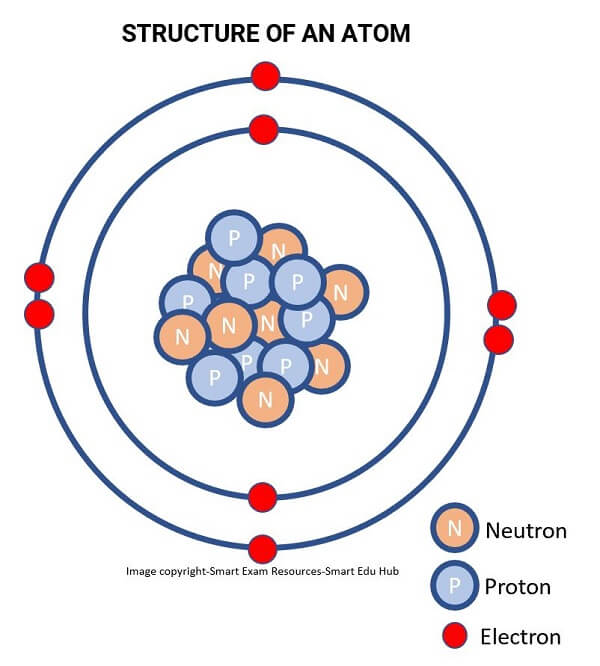
Structure of an atom
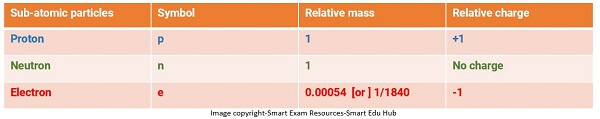
Define:
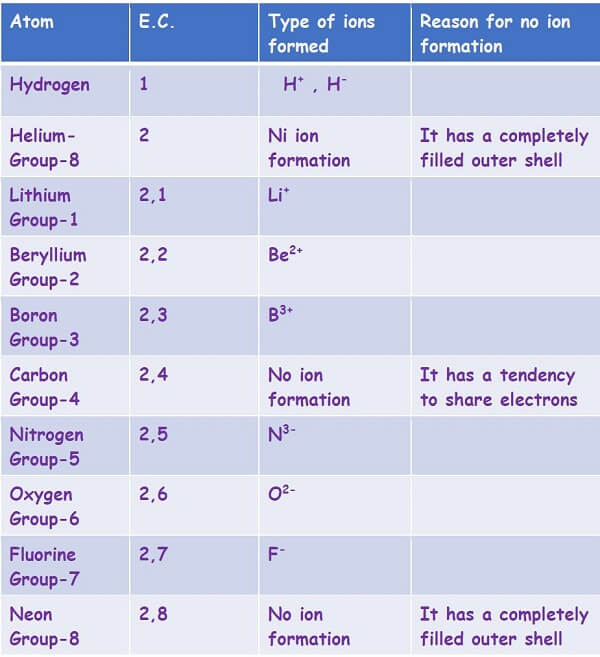
The above video is a preview of the actual revision notes available as a part of the paid membership. All learning resources in the form of pdfs that are available on the website are 100% printable and downloadable.
Click the highlighted blanks to reveal the correct answers.
Unlock the complete range of topic questions, solved past exam papers along with mock exams, detailed and downloadable revision notes, timed quizzes, interactive flipbooks and many more exam preparation resources.
Unlock Complete Resources
- Smart Exam Resources

Write a public review