Definition and examples
Isotopes are different atoms of the same element that have the same number of protons but different numbers of neutrons.
Examples of some isotopes
| Elements | Isotopes |
|---|---|
| Chlorine | 3517Cl 3717Cl |
| Carbon | 126C 136C |
| Hydrogen | 11H 21H 31H |
Isotopes are different atoms of the same element that have the same number of protons but different numbers of neutrons.
| Elements | Isotopes |
|---|---|
| Chlorine | 3517Cl 3717Cl |
| Carbon | 126C 136C |
| Hydrogen | 11H 21H 31H |
3517Cl?
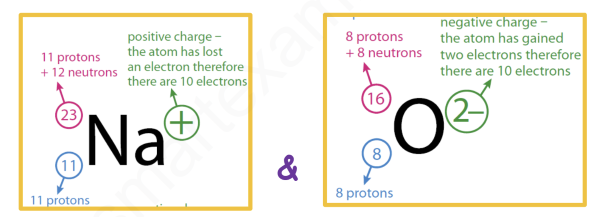
2412Mg2+
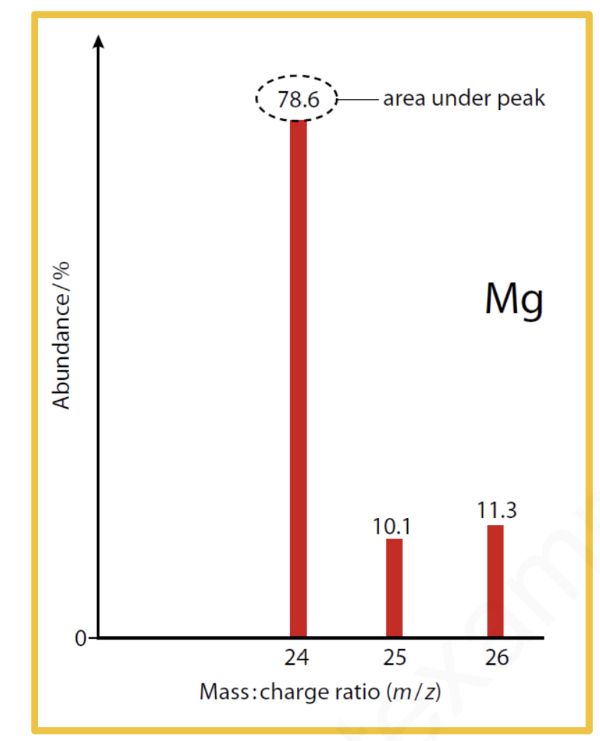
Note: The scale on the x-axis is m/z, which represents the mass:charge . we can also consider this to be the mass of the isotope. [ Detailed explanation has not been provided as it is not required as per the syllabus].
EXTENDED:
The above video is a preview of the actual revision notes available as a part of the paid membership. All learning resources in the form of pdfs that are available on the website are 100% printable and downloadable.
IGCSE Chemistry Revision Interactive Notes Isotopes
Topic: Atoms, elements and compounds
Click the highlighted blanks to reveal the correct answers.
Unlock the complete range of solved past exam papers along with mock exams, revision notes, timed quizzes, interactive flipbooks and many more exam preparation resources.
Unlock Complete Resources
Write a public review